Spectral Resolution Add-on Device for Two Photon Microscope

OTT1105/1242
Applications
Imaging of Live Cell, 3D, Deep Tissue, and Long-Term without compromised tissue viability, Whole Organ or Slice Imaging
Target Problems
- Typical two photon microscopes scramble dynamic spectral information in living cells
- Sample scanning limitations and spectral bleed severely limit the imaging capabilities in most microscopes
Key Features
- Single Scan Capture –No need for multiple scans to resolve spectrum and no photobleaching
- Easy Installation (Add-on Device)–Be integrated into variety of microscopes with fluorescence and either brightfield or phase contrast
- High Resolution –Delivers high spatial and spectral resolutionin real time
- Unsurpassed Detection Capability –Provides spectral information up to 100x magnification
- Speed and Sensitivity –Has the sensitivity and speed of competing systems, and is similar to that of broadband microscopes
- Wide Wavelength Range –Large numbers of fluorescent markers are easily resolvable spectrally
- Proven Technology –Based on well-established quantitative FRET imaging
Technology
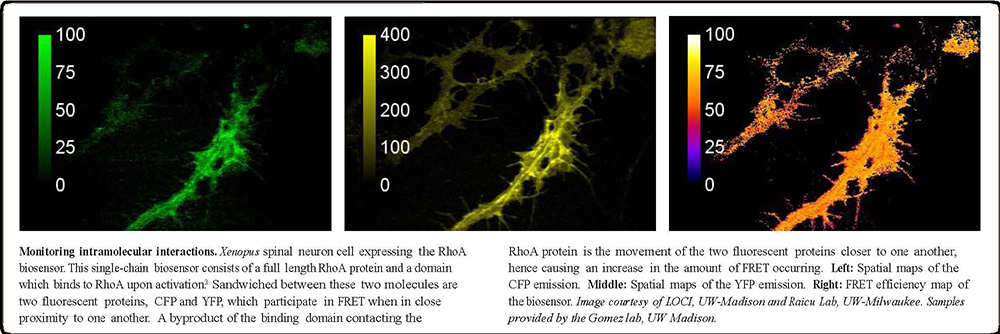
The add-on device may be attached to the existing research microscope via a side port that incorporates a femtosecond laser (as shown in the picture). This state-of-the-art imaging tool uses a single scan multiphoton excitation (425 to 650 nm wavelength range) to deliver pixel level spectral resolution of complex, multi-color fluorescence samples. This technology can upgrade the imaging capability of a variety of microscopes, enabling it to achieve multi-photon microscope grade with 3D spatial resolution. This is the only scanning system that permits real-time quantitative analysis of a single molecule, molecular complexes, and their spatial distribution in living cells. This technology also allows fluorescence signals from co-localized molecules in a three-dimensional sample to be resolved spectrally by parallel detection of tens or hundreds of wavelengths using an EMCCD camera without the need of Confocal setup.
Intellectual Property
US7,973,927 | US8,094,304 | US9,103,721 | US8,982,206 | EP20120782796 validated in BE, FR, DE, UK, and IE.
Inventor
Dr. Valerica Raicu, Professor of Physics at the University of Wisconsin-Milwaukee